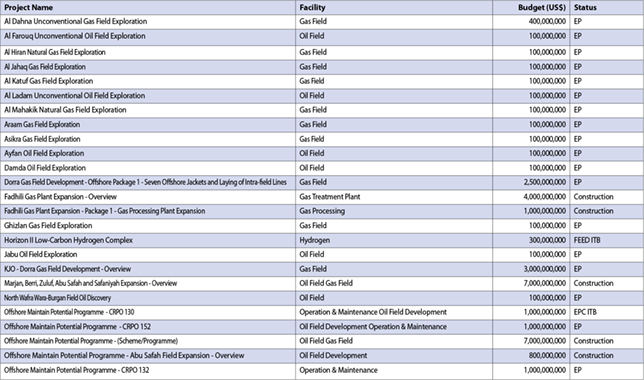
Varnish removal and prevention using EPT Clean Oil systems
Varnish removal and prevention using EPT Clean Oil systems
An understanding of lubricant solvency is the key to understanding the mechanism by which varnish deposits are formed and, more importantly, the mechanism by which they can be removed, Peter Dufresne of EPT Clean Oil, tells OGN
Varnishing greatly affects the availability and reliability of turbines, integral to the power generation industry.
The primary reason that varnish continues to be problematic is that the mechanism by which varnishing occurs is poorly understood.
An understanding of lubricant solvency is the key to understanding the mechanism by which varnish deposits are formed and, more importantly, the mechanism by which they can be removed.
According to ‘A Comprehensive Guide to Industrial Turbine Lubricants’, by Afton Chemical Corp, oil problems are estimated to be the cause of nearly one-fifth of all forced turbine outages.
THE VARNISH CYCLE
The typical varnish formation cycle in a gas turbine involves the following steps:
 |
Figure 2 ... lubricants made with Group II are oxidatively stable than |
• Oxidation is a chemical reaction between the lubricant base stock and oxygen in the air. Oxidation is unavoidable and begins to take place the instant that a new fluid is exposed to air for the first time, regardless of whether or not the fluid is put into service.
Like all other chemical reactions, the rate of oxidation is bound by the Arrhenius equation, which states that the rate of reaction will double for every temperature increase of 10 deg C (18 deg F).
Once a new fluid is put into service, it is exposed to greater temperatures and experiences a concomitant increase in the rate at which it oxidises.
Even when operating temperatures are a typical 50 deg C (125 deg F), bearings may reach temperatures in excess of 150 deg C (300 deg F); the rate of oxidation at the bearing in this instance will be 1,024 (210) times greater than that in the cooler regions of the system. As a result, oxidation typically occurs wherever hot spots are found.
 |
Figure 1: Successful varnish mitigation strategies |
Oxidation products build up in the lubricant over time, but remain dissolved at operating temperatures unless they exceed the fluid’s saturation point.
• As the oil moves from hotter regions within the system to cooler ones (hydraulic lines supplying high pressure oil to engine geometry control, for instance), the fluid temperature falls and the solubility of any varnish precursors present decreases. These precursors begin to precipitate from solution in the form of particulates.
Like water freezing to form ice, this precipitation of varnish is a physical change and not a chemical reaction.
• Once formed, varnish particulates agglomerate and form deposits, preferentially coating metal surfaces. These deposits are often the cause of unit trips or fail-to-start conditions. Like precipitation in step 2 above, agglomeration and deposition are physical changes.
This model of varnish formation is widely accepted and reasonably well-understood. What is less well-understood, however, is the fact that once varnish deposits form, they can be reabsorbed if the solvency of the lubricant is increased.
While the chemical changes that lead to the formation of varnish precursors (step 1) are irreversible, the physical changes (steps 2 and 3) which led to the formation of varnish deposits are reversible. Successful varnish mitigation strategies use this fact to their advantage (figure 1).
STRATEGIES TO COMBAT VARNISHING
Most modern turbine lubricants are made with API (American Petroleum Institute) Group II mineral oil base stocks, which contain an anti-oxidant additive package. The chemistry of Group II base stocks makes them more oxidatively stable than the traditional Group I base stocks (figure 2).
Antioxidants are added to the lubricant as a built-in varnish mitigation strategy. Antioxidants additives generally comprise two classes of chemicals: phenols and amines. These chemical additives are sacrificial and will oxidise more readily than the oil base stock.
Unfortunately, both phenols and amines are consumed as they oxidise. Once the additives are consumed, the rate of fluid degradation accelerates, returning to that of the non-additised base stock (figure 2). Antioxidants limit the rate of oxidative degradation and, therefore, delay varnishing, but they cannot prevent it.
When the lubricant inevitably oxidises and varnish precursors are formed, varnish removal systems are necessary to prevent degradation products from accumulating to the point where varnishing occurs.
There are two main types of varnish removal systems: those based on the removal of suspended particles and those based on the removal of soluble varnish and its precursors.
• Suspended particle removal systems: Depth filtration, Balanced Charge Agglomeration (BCA™), electrostatic oil cleaning or combinations of these techniques are advanced forms of particulate removal.
Since solvency decreases at lower temperatures (favouring the formation of particulates), the maximum benefit using these systems is achieved when the turbine is not operating and the lubricant is at ambient temperatures. Therefore, these systems are of more use when employed periodically, during outages; they are less effective when used continuously during turbine operation.
When used during normal turbine operation, these systems are incapable of removing soluble varnish and its precursors.
To overcome this limitation, oil coolers can be used to accelerate the varnish formation cycle and precipitate particulate from the lubricant immediately before it passes through the varnish removal system. However, cool oil is more viscous making it difficult to pass through these filtration systems.
The oil cannot be cooled to the temperatures required for complete removal of all soluble varnish. Therefore, lubricant solvency is never improved to the point where existing varnish deposits can be re-dissolved into the fluid.
• SVR™ lubricant conditioning system: SVR is an engineered lubricant conditioning system that targets the underlying cause of lubricant failure: chemical breakdown.
Utilising patented ion-exchange technology, ICB™, soluble varnish and its precursors are adsorbed and removed by billions of polar sites. This adsorption relies on a preferential molecular interaction between the polar varnish molecules and the polar sites present within the resin.
Just as polar varnish prefers to coat polar metal surfaces, so too it prefers to adsorb on the polar sites of the ICB resin.
Conventional ion exchange resins function by exchanging one chemical for another. Unlike these resins which exchange one contaminant for another, ICB resins are engineered to adsorb the entire contaminant without returning any others to the fluid.
A key benefit of the ICB adsorption principle is that harmful oxidation products can be removed at any operating temperature, meaning that SVR systems can be used continuously.
The continuous removal of soluble varnish and its precursors ensures that degradation products do not accumulate in the lubricant, eliminating the risk of varnish formation during normal turbine shut down cycles.
Moreover, the continuous removal of soluble varnish produces a lubricant with extremely high solvency.
Since the physical changes that resulted in the formation of insoluble varnish particles and deposits are reversible, the high solvency of the SVR-treated lubricant forces insoluble varnish already present on turbine surfaces back into the soluble varnish form where they can be adsorbed and removed. With all the remaining oxidation by products removed, the varnish formation cycle is completely stopped.
SUMMARY
Varnish particles and deposits are created from reversible physical changes that begin with soluble oxidation products and end with varnish deposits. For these changes to be reversible, the chemistry of the deposits has to be similar to the chemistry of the lubricant from which the deposits originated.
Once fluid solvency has been increased (by removing soluble varnish at normal operating temperature), deposits will simply dissolve back into the fluid and be removed.